ΟΙ ΕΠΙΔΡΑΣΕΙΣ ΤΟΥ ΥΨΗΛΑ ΦΑΙΝΟΛΙΚΑ ΕΞΑΙΡΕΤΙΚΟΥ ΠΑΡΘΕΝΟΥ ΕΛΑΙΟΛΑΔΟY ELETTRA ΣΤΟ ΧΡΟΝΙΟ ΠΟΝΟ ΤΗ ΦΛΕΓΜΟΝΗ ΚΑΙ ΤΗΝ ΟΣΤΕΟΑΡΘΡΙΤΙΔΑ

Κλινική μελέτη
Πραγματοποιήθηκε από τον Charan Chadha
Οι Επιδράσεις του Υψηλά Φαινολικά Εξαιρετικού Παρθένου Ελαιόλαδου ELETTRA στον Χρόνιο Πόνο, τη Φλεγμονή και την Οστεοαρθρίτιδα

Σχετικά με τις ελιές και το ελαιόλαδο
Η ελιά (Olea Europaea) είναι ένα δέντρο με βρώσιμους καρπούς, φύλλα και σπόρους. Το ελαιόλαδο προέρχεται από τον καρπό της ελιάς και περιέχει μονοακόρεστα λιπαρά οξέα. Τα λιπαρά οξέα στο ελαιόλαδο φαίνεται να μειώνουν τα επίπεδα χοληστερόλης και έχουν αντιφλεγμονώδη δράση. Το ελαιόλαδο χρησιμοποιείται συνήθως σε τρόφιμα.
Ως φάρμακο, οι άνθρωποι χρησιμοποιούν συνήθως ελαιόλαδο για καρδιακές παθήσεις, διαβήτη και υψηλή αρτηριακή πίεση. Χρησιμοποιείται επίσης για υψηλή χοληστερόλη, καρκίνο, δεξιότητες μνήμης και σκέψης, ημικρανία, παχυσαρκία και πολλές άλλες καταστάσεις, αλλά δεν υπάρχουν καλά επιστημονικά στοιχεία που να υποστηρίζουν πολλές από αυτές τις άλλες χρήσεις.
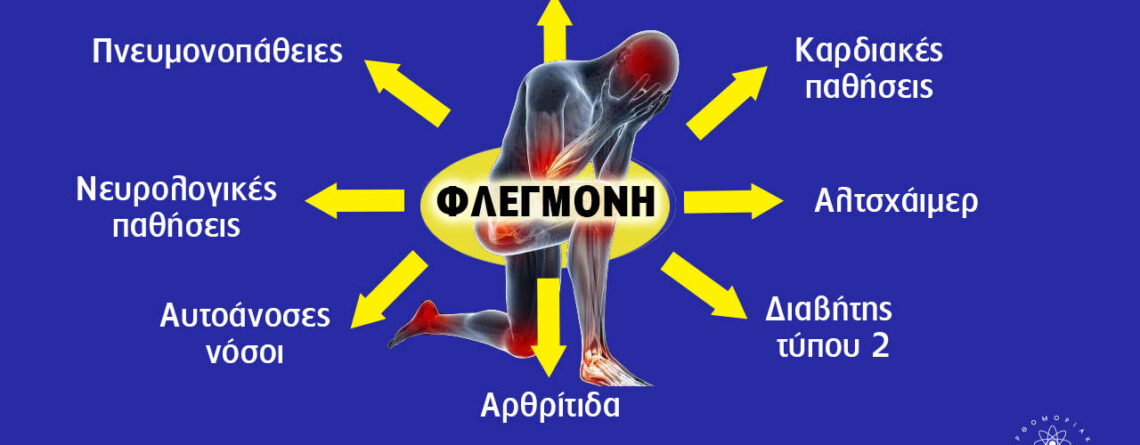
Κατά τη διάρκεια του 2021, εκτιμάται ότι το 20,9% των ενηλίκων των ΗΠΑ (51,6 εκατομμύρια άτομα) παρουσίασαν χρόνιο πόνο, και το 6,9% (17,1 εκατομμύρια άτομα) παρουσίασαν χρόνιο πόνο υψηλής επίπτωσης (δηλαδή, χρόνιο πόνο που οδηγεί σε ουσιαστικό περιορισμό στις καθημερινές δραστηριότητες).
Σκοπός
Αυτή η μελέτη έγινε για να καταλάβουμε εάν μπορούμε να χρησιμοποιήσουμε μια πηγή «τροφής» με υψηλές φαινόλες για να βοηθήσουμε μείωση του πόνου και της φλεγμονής, ενώ παράλληλα βελτιώσει τη λειτουργία και τον τρόπο ζωής χωρίς όλες τις παρενέργειες που έχουν τα συνταγογραφούμενα και μη συνταγογραφούμενα φάρμακα.

Μερικά στατιστικά στοιχεία για το πλαίσιο
Ο χρόνιος πόνος και η φλεγμονή είναι ένα διάχυτο μέρος του συστήματος υγειονομικής περίθαλψής μας και όσοι πάσχουν από αυτές τις παθήσεις συχνά αναζητούν ιατρική φροντίδα από διάφορους πόρους, όπως ενδεικτικά γιατρούς, χειροπράκτες, φυσιοθεραπευτές, θεραπευτές μασάζ και άλλους επαγγελματίες υγείας. Συχνά αντιμετωπίζονται με μια σειρά συνταγογραφούμενων φαρμάκων, φάρμακα χωρίς ιατρική συνταγή, ενέσεις, χειρουργικές επεμβάσεις και ούτω καθεξής.
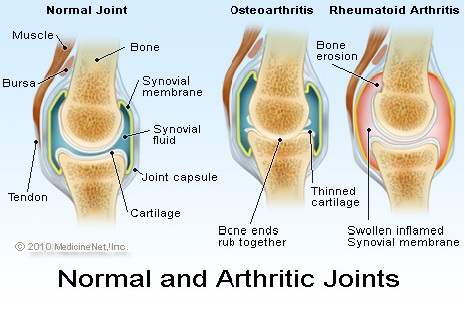
Οστεοαρθρίτιδα
Με βάση τα δεδομένα από την Εθνική Έρευνα Συνεντεύξεων Υγείας (NHIS), κατά την περίοδο 2016 – 2018, εκτιμάται ότι 58,5 εκατομμύρια ενήλικες στις ΗΠΑ (23,7%) είχαν ποτέ ενημερωθεί από γιατρό (με διάγνωση γιατρού) ότι είχαν κάποια μορφή αρθρίτιδας, ρευματοειδούς αρθρίτιδας, ουρικής αρθρίτιδας, λύκου ή ινομυαλγίας.
• Μέχρι το 2040, υπολογίζεται ότι 78,4 εκατομμύρια (25,9%) ενήλικες στις ΗΠΑ ηλικίας 18 ετών και άνω αναμένεται να έχουν αρθρίτιδα με διάγνωση γιατρού (BRFSS).4
• Η αρθρίτιδα περιορίζει τις δραστηριότητες 25,7 εκατομμυρίων ενηλίκων στις ΗΠΑ. Περίπου το 44% των ενηλίκων με αρθρίτιδα που διαγνώστηκε από γιατρό είχε περιορισμούς δραστηριότητας που αποδίδονται στην αρθρίτιδα κατά την περίοδο 2016 – 2018 (NHIS).1
• Το 2013, το εθνικό κόστος της αρθρίτιδας ήταν συνολικά 304 δισεκατομμύρια δολάρια.8
• Το ιατρικό κόστος που αποδίδεται στην αρθρίτιδα ήταν 140 δισεκατομμύρια δολάρια.8
• Οι χαμένοι μισθοί που αποδίδονται στην αρθρίτιδα ήταν 164 δισεκατομμύρια δολάρια.
Επικράτηση ανά ηλικία
• Η αρθρίτιδα επηρεάζει ενήλικες όλων των ηλικιών και ο επιπολασμός της αρθρίτιδας αυξάνεται με την ηλικία. Από το 2016 έως το 2018 στις Ηνωμένες Πολιτείες (NHIS):
• Το 7,1% των ενηλίκων 18 έως 44 ετών ανέφερε αρθρίτιδα.1
• Μεταξύ των ενηλίκων ηλικίας 45 έως 64 ετών, σχεδόν το ένα τρίτο (30,5%) ανέφερε αρθρίτιδα που διαγνώστηκε από γιατρό.1
• (50,4%) ανέφερε αρθρίτιδα με διάγνωση γιατρού.1
Πτώσεις και τραυματισμοί πτώσης
Οι πτώσεις και οι τραυματισμοί από πτώση αποτελούν σημαντική ανησυχία για ενήλικες με αρθρίτιδα.
• Το 2012, σε σύγκριση με τους ενήλικες ηλικίας 45 ετών και άνω που δεν έχουν αρθρίτιδα, οι ενήλικες 45 ετών και άνω με αρθρίτιδα ήταν (BRFSS):
• 4 φορές περισσότερες πιθανότητες να έχουμε δύο ή περισσότερες πτώσεις.7 • 5 φορές περισσότερες πιθανότητες τραυματισμού από πτώση.7
Στοιχεία από την Εθνική Έρευνα Εξέτασης Υγείας και Διατροφής
Την περίοδο 2015–2018, το 10,7% των ενηλίκων των ΗΠΑ ηλικίας 20 ετών και χρησιμοποίησαν υπερβολικά ένα ή περισσότερα συνταγογραφούμενα παυσίπονα (οπιούχα ή μη) τις τελευταίες 30 ημέρες.
Την περίοδο 2015–2018, το 5,7% των ενηλίκων των ΗΠΑ χρησιμοποίησε ένα ή περισσότερα συνταγογραφούμενα οπιοειδή. Η συνταγογραφούμενη χρήση οπιοειδών ήταν υψηλότερη στις γυναίκες παρά στους άνδρες και η χρήση αυξανόταν με την ηλικία.
Το 2015–2018, το 5,0% των ενηλίκων χρησιμοποίησε μη οπιοειδή συνταγογραφούμενα παυσίπονα χωρίς συνταγογραφούμενα οπιοειδή τις τελευταίες 30 ημέρες. Η χρήση ήταν η χαμηλότερη μεταξύ των ενηλίκων μη Ισπανόφωνων Ασιατών.
Από το 2009-2010 έως το 2017-2018, δεν παρατηρήθηκε σημαντική αλλαγή στη χρήση συνταγογραφούμενων οπιοειδών, αλλά η χρήση μη οπιοειδών συνταγογραφούμενων παυσίπονων (χωρίς συνταγογραφούμενα οπιοειδή) αυξήθηκε.
Συνταγογραφούμενα παυσίπονα
χρησιμοποιούνται για τη θεραπεία του πόνου λόγω τραυματισμού, χειρουργικής επέμβασης και παθήσεων υγείας, όπως η αρθρίτιδα και ο καρκίνος. Ενώ τα οπιοειδή μπορεί να συνταγογραφούνται μαζί με μη οπιοειδή παυσίπονα, οι μη φαρμακολογικές και οι φαρμακολογικές θεραπείες που δεν περιέχουν οπιοειδή προτιμώνται για τη διαχείριση του χρόνιου πόνου, όπου χρειάζεται.9
Αυτή η αναφορά δείχνει το ποσοστό των ενηλίκων των ΗΠΑ που, τις τελευταίες 30 ημέρες, χρησιμοποίησαν ένα ή περισσότερα συνταγογραφούμενα παυσίπονα, χρησιμοποίησαν συνταγογραφούμενα οπιοειδή φάρμακα ή χρησιμοποίησαν μη συνταγογραφούμενα φάρμακα παυσίπονα (χωρίς συνταγογραφούμενα οπιοειδή) το 2015–2018.
Σε μια μελέτη με περισσότερους από 340 ασθενείς που έπασχαν από πόνο στη μέση ή στον αυχένα, μια ομάδα Αυστραλών ερευνητών διαπίστωσε ότι δεν υπήρχε διαφορά στον πόνο βαρύτητα μετά από έξι εβδομάδες μεταξύ εκείνων που έλαβαν οπιοειδή έναντι ενός χαπιού ζάχαρης εικονικού φαρμάκου. Τα Κέντρα Ελέγχου και Πρόληψης Νοσημάτων (CDC) συνιστούν στους επαγγελματίες να μην συνταγογραφούν οπιοειδή ως θεραπεία πρώτης γραμμής για τον χρόνιο πόνο στην πλάτη.
Το CDC συνιστά οι πάροχοι υγειονομικής περίθαλψης να θεραπεύουν αρχικά ασθενείς με χρόνιο πόνο στην πλάτη ή τον αυχένα χρησιμοποιώντας μη φαρμακευτικές θεραπείες ή άλλα φάρμακα εκτός των οπιοειδών και τα οπιοειδή θα πρέπει να χρησιμοποιούνται μόνο εάν τα αναμενόμενα οφέλη τόσο για τον πόνο όσο και για τη λειτουργία αναμένεται να αντισταθμίσουν τους κινδύνους για τον ασθενή.
Η μελέτη αναφέρει επίσης ότι οι ανασκοπήσεις της ιατρικής βιβλιογραφίας βρήκαν «ελάχιστα στοιχεία» ότι τα οπιοειδή είναι αποτελεσματικά για τη θεραπεία του χρόνιου πόνου στην πλάτη. Επιπλέον, η μελέτη έδειξε ότι για όλους τους τύπους μη καρκινικού πόνου — ο οποίος περιλαμβάνει, αλλά δεν περιορίζεται σε, τον αυχένα και πόνος στην πλάτη — η αποτελεσματικότητα των οπιοειδών είναι περίπου 30% για βραχυπρόθεσμη ανακούφιση από τον πόνο και δεν βελτίωσε τη σωματική λειτουργία.
Παρενέργειες Φαρμάκων
Μπορεί να ποικίλλει ανάλογα με το κάθε άτομο. Με βάση αναφορές (αν και όχι αποκλειστικά), έχουν εντοπιστεί τα ακόλουθα συμπτώματα:
• Κνησμός • Δυσκοιλιότητα • Κυψέλες • Έλκος • Εξάνθημα • Έμετος • Πρήξιμο στο στόμα ή στο πρόσωπο • Καταστολή • Δυσκολία στην αναπνοή • Αυξημένος κίνδυνος για πτώσεις και κατάγματα • Διαταραγμένος ύπνος • Αρκετοί άλλοι • Ναυτία
Μια μελέτη του 2015 αναφέρει ότι ενώ τα οπιοειδή είναι τα πιο συνταγογραφούμενα παυσίπονα στις ΗΠΑ (με περισσότερους από τους μισούς τακτικούς χρήστες οπιοειδών να αντιμετωπίζουν πόνο στην πλάτη), δεν έχει αποδειχθεί ότι βοηθούν τους ανθρώπους να επιστρέψουν στην εργασία τους πιο γρήγορα, ούτε βελτιώνουν τη λειτουργία τους όταν χρησιμοποιούνται για τη θεραπεία ένα οξύ επεισόδιο.2
Θέματα Μελέτης και Ιατρικό Πρωτόκολλο
Όταν λαμβάνεται από το στόμα
Όταν λαμβάνεται από το στόμα, το ελαιόλαδο καταναλώνεται συνήθως στα τρόφιμα. Έως και ένα λίτρο έξτρα παρθένου ελαιόλαδου την εβδομάδα χρησιμοποιείται με ασφάλεια ως μέρος μιας μεσογειακής διατροφής για έως και 5,8 χρόνια. Το ελαιόλαδο είναι συνήθως καλά ανεκτό. Μπορεί να προκαλέσει ναυτία σε μικρό αριθμό ατόμων. Ως υπενθύμιση σε όλους, σκοπός αυτής της μελέτης ήταν να κατανοήσουμε τα οφέλη των φαινολών (Συγκεκριμένα Η ελαιοκανθάλη μαζί με άλλες που βρίσκονται στις ελιές) στον πόνο και τη φλεγμονή καθώς σχετίζονται με την απώλεια λειτουργικότητας.
Η μελέτη διεξήχθη σε περίοδο 30 ημερών και περιελάμβανε αρχικά τριάντα ειδικευμένα άτομα. Κανένα από τα τριάντα άτομα δεν είχε αναφέρει ότι έπαιρνε ελαιόλαδο υψηλής περιεκτικότητας σε φαινολικά συστατικά στο παρελθόν για οποιονδήποτε λόγο.
Το πρωτόκολλο περιελάμβανε τα εξής:
1. Αρχική αναφορά πόνου (σε αναλογική κλίμακα 0-10) Όλα τα άτομα έπρεπε να έχουν ελάχιστο πόνο 4/10 και μέγιστο 9/10.
2. Όλα τα άτομα έπρεπε να υποβληθούν σε ιατρική διάγνωση χρόνιου πόνου ή οστεοαρθρίτιδας, μαζί με τις αρχικές παραμέτρους όπως ορίστηκαν πριν από την έναρξη της μελέτης.
3. Όλα τα άτομα είχαν αναφέρει απώλεια λειτουργικότητας σε καθημερινές δραστηριότητες συμπεριλαμβανομένων, ενδεικτικά, (περπάτημα, αναρρίχηση/κάθοδος σκαλοπατιών, γκολφ, τένις, ύπνος και ούτω καθεξής).
4. Τα υποκείμενα έπρεπε να διακόψουν όλα τα παυσίπονα πριν από την έναρξη της μελέτης για τουλάχιστον 3 ημέρες πριν από τη μελέτη. Αυτό έγινε για να καθαριστεί το σύστημα του σώματος από φάρμακα που θα μπορούσαν να επηρεάσουν τα αποτελέσματα του ελαιολάδου με υψηλή περιεκτικότητα σε φαινολικά συστατικά, όπως σχετίζεται με αυτήν τη μελέτη.
5. Όλα τα άτομα ακολούθησαν το πρωτόκολλο των 2 κουταλιών της σούπας (TBSP) κάθε πρωί χωρίς να φάνε για 30 λεπτά. Αυτό το πρωτόκολλο δημιουργήθηκε από επιστήμονες για το World Olive Center for Health.
6. Δόθηκαν εβδομαδιαίες αναφορές για τη μελέτη του ερευνητή που περιγράφουν τις αλλαγές (αν υπάρχουν) στον πόνο και/ή στη λειτουργία. Αυτό θα περιλαμβάνει επίσης τυχόν άλλα ευρήματα που ήταν «παρενέργειες».

ΑΠΟΤΕΛΕΣΜΑΤΑ
Τριάντα άτομα επιλέχθηκαν για τη μελέτη.
Υπήρχαν είκοσι έξι γυναίκες και 4 άνδρες που πληρούσαν τις προϋποθέσεις για τη μελέτη. Οι ηλικίες κυμαίνονταν μεταξύ 22-76 ετών.
Δύο (2) άτομα αποκλείστηκαν κατά τη διάρκεια της μελέτης για διάφορους λόγους, καθώς σχετίζονται με τις απαιτήσεις του πρωτοκόλλου και ένα άτομο υπέστη τραυματισμό που απαιτούσε συνταγογραφούμενη φαρμακευτική αγωγή για τη διαχείριση του οιδήματος. Οι τελικές εκθέσεις περιλάμβαναν είκοσι επτά άτομα.
Για τους σκοπούς της μελέτης οι τελικές αναφορές περιελάμβαναν επίπεδα πόνου και λειτουργικές βελτιώσεις με βάση τα αρχικά ελλείμματα. Τα παρακάτω ποσοστά βασίζονται στο τελικό μέγεθος δείγματος των είκοσι επτά.
• Είκοσι πέντε άτομα (93%)
Αναφέρθηκαν σε σημαντικές διαφορές στα επίπεδα πόνου και/ή βελτιωμένη λειτουργία. Όλα τα άτομα που μίλησαν για μείωση του πόνου ανέφεραν ότι δεν υπήρχε ανάγκη λήψης πρόσθετων παυσίπονων. Ανέφεραν μάλιστα ότι ρυθμίστηκε το πεπτικό τους σύστημα.
• Τρία άτομα (11%) Ανέφεραν μείωση της εμμηνορροϊκής κράμπας από την οποία υπέφεραν σε όλη την ενήλικη ζωή τους.
• Έντεκα άτομα (41%) Ανέφεραν αυξημένη βελτίωση του ύπνου αναφέροντας τα ακόλουθα παραδείγματα (Περισσότερος ύπνος, βαθύτερος ύπνος, λιγότερος αριθμός αφυπνίσεων λόγω μείωσης του πόνου).
• Δεκαέξι άτομα (59%) ευκολότερες και συχνότερες κενώσεις. Λιγότερο φούσκωμα, μειωμένα συμπτώματα παλινδρόμησης.
• Δύο άτομα (7%) Ανέφεραν ότι δυσκολεύονταν να ανεχθούν την κατάποση των 2 κουταλιών ελαιολάδου, αλλά ήταν σε θέση να ολοκληρώσουν το πρωτόκολλο.
• Ένα άτομο (3%) Ανέφερε ναυτία κατά τη λήψη ελαιολάδου με υψηλή περιεκτικότητα σε φαινολικά συστατικά.
• Δύο άτομα (7%) Δεν αναφέρθηκε καμία αλλαγή στα επίπεδα πόνου, αλλά διαπίστωσαν αύξηση της λειτουργικότητας τους.
Μελλοντικές Μελέτες και Συμπέρασμα
Όπως μπορούν να επιβεβαιώσουν οι περισσότεροι Αμερικανοί με χρόνιο πόνο και φλεγμονή ή/και οστεοαρθρίτιδα, τα επίπεδα πόνου και η απώλεια λειτουργικότητας τους αναγκάζουν να αναζητήσουν ιατρική φροντίδα που μπορεί και συχνά περιλαμβάνει συνταγογραφούμενα/μη συνταγογραφούμενα φάρμακα, ενέσεις, χειρουργικές επεμβάσεις καθώς και άλλες εναλλακτικές θεραπείες.
Αυτή η μελέτη διεξήχθη για να κατανοηθεί εάν υπάρχουν οφέλη από τη χρήση ελαιολάδου υψηλής περιεκτικότητας σε φαινολικά ως υποκατάστατο συνθετικών ή φαρμακευτικών φαρμάκων για τη διαχείριση του χρόνιου πόνου/φλεγμονής ή της οστεοαρθρίτιδας.
Όπως σημειώθηκε παραπάνω, υπάρχουν ισχυρά στατιστικά στοιχεία που δείχνουν ότι τα οφέλη του υψηλού φαινολικού ελαιολάδου μπορεί να μειώσουν τα επίπεδα πόνου και να βελτιώσουν τη λειτουργία και τον τρόπο ζωής των ανθρώπων που υποφέρουν από αυτές τις καταστάσεις.

Μελλοντικές Σπουδές
Καθώς υπήρχαν αρκετές θετικές παρενέργειες, καθώς σχετίζονται με τη βελτίωση του ύπνου και τις κράμπες της περιόδου αναφέρθηκαν και άλλα πρόσθετα οφέλη που θα διερευνήσει ο ερευνητής σε μελλοντικές μελέτες.
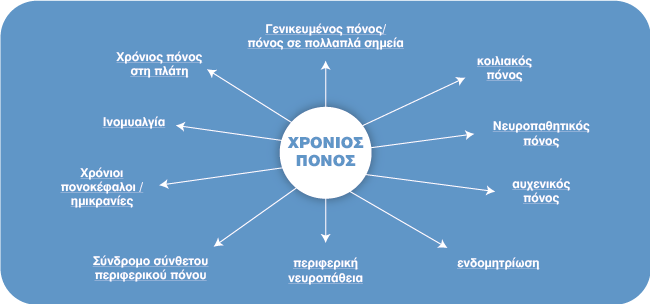
Περιορισμοί της Μελέτης
Καθώς οι περισσότερες μελέτες μπορεί να περιλαμβάνουν έλεγχο και/ή εικονικό φάρμακο- αυτή η μελέτη ΔΕΝ χρησιμοποίησε εικονικό φάρμακο ή διπλά τυφλή.
Ο ερευνητής έψαχνε για συγκεκριμένα θέματα που μπορεί να ωφεληθούν από το (HPEVOO). Αυτή η μελέτη δεν καθιέρωσε φάρμακα για τον πόνο πριν ή κατά τη διάρκεια της μελέτης, έτσι ώστε η μόνη μεταβλητή να ήταν το αντικείμενο της μελέτης (Υψηλό Φαινολικό Ελαιόλαδο).
Εάν τα άτομα έπαιρναν φάρμακα προηγουμένως, τα διέκοψαν για τουλάχιστον 3 ημέρες πριν από τη μελέτη. Η μελέτη ΔΕΝ πραγματοποιήθηκε ως τυχαιοποιημένη διπλή-τυφλή μελέτη. Για τους σκοπούς της κλινικής δοκιμής, θα ήταν ωφέλιμο να πραγματοποιηθεί μια τυχαιοποιημένη διπλή-τυφλή μελέτη για την πλήρη αναγνώριση των οφελών ή διαφορές μεταξύ ιατρικών παρεμβάσεων. Η μελέτη διεξήχθη σε τριάντα άτομα. Αν και ήταν ένα καλό αρχικό μέγεθος δείγματος, η επόμενη φάση της μελέτης θα πραγματοποιηθεί σε μεγαλύτερο μέγεθος δείγματος.
1. Theis KA, Murphy LB, Guglielmo D, et al. Prevalence of Arthritis and arthritis-attributable activity limitation — United States, 2016–2018. MMWR Morb Mortal Wkly Rep 2021;70:1401–1407. doi: http://dx.doi.org/10.15585/mmwr.mm7040a2 2. Duca LM, Helmick CG, Barbour KE, et al. State-specific prevalence of inactivity, self-rated health status, and severe joint pain among adults with arthritis — United States, 2019. Prev Chronic Dis 2022;19:210346. doi: http://dx.doi.org/10.5888/pcd19.210346 3. Barbour KE, Moss S, Croft JB, et al. Geographic variations in arthritis prevalence, health-related characteristics, and management — United States, 2015. MMWR Surveill Summ 2018;67(No. SS-4):1–28. doi: http://dx.doi.org/10.15585/mmwr.ss6704a1. 4. Hootman JM, Helmick CG, Barbour KE, Theis KA, Boring MA. Updated projected prevalence of self-reported doctor-diagnosed arthritis and arthritis-attributable activity limitation among US adults, 2015–2040. Arthritis & Rheumatol. 2016;68(7):1582–1587. doi: 10.1002/art.39692. PubMed PMID: 27015600. 5. Barbour KE, Boring M, Helmick CG, Murphy LB, Qin J. Prevalence of severe joint pain among adults withdoctor-diagnosed arthritis — United States, 2002–2014. MMWR Morb Mortal Wkly Rep 2016;65:1052–1056. doi: http://dx.doi.org/10.15585/mmwr.mm6539a2. 6. Theis KA, Roblin D, Helmick CG, Luo R. Prevalence and causes of work disability among working-age US adults: 2011–2013. Disabil Health J. 2018;11(1):108–115. doi: 10.1016/j.dhjo.2017.04.010. PMID: 28476583. 7. Barbour KE, Stevens JA, Helmick CG, et al. Falls and fall injuries among adults with arthritis—United States, 2012. Morb Mortal Wkly Rep.2014;63(17):379-383. 8. Murphy LB, Cisternas MG, Pasta DJ, Helmick CG, Yelin EH. Medical expenditures and earnings losses among US adults with arthritis in 2013. Arthritis Care Res (Hoboken). 2018;70(6):869-876. doi: 10.1002/acr.23425. 9. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. MMWR Recomm Rep 65(1):1–49. 2016.
